|
4/3/2024 0 Comments Daltons atomic theoryThe law of conservation of mass states that the total mass before and after a chemical reaction remains constant. During the formation of water, existing hydrogen and oxygen atoms are neither created nor destroyed, they only rearrange.ĭalton built his atomic theory upon two previous laws of chemical reactions: the law of conservation of mass and the law of definite proportions. The last postulate of Dalton’s theory states that chemical reactions do not change the atoms of one element into atoms of a different element instead, the existing atoms rearrange themselves to form new substances. For example, two atoms of hydrogen and one atom of oxygen combine in a 2 to 1 ratio to form H 2O - water. The third postulate states that atoms of different elements may combine with each other, in simple whole-number ratios, to form compounds. Thus, all the atoms of hydrogen are identical to each other, but differ from oxygen atoms. The second postulate states that while atoms of the same element are identical, they differ from the atoms of other elements. For example, hydrogen is made up of two hydrogen atoms bonded together and oxygen is made up of two oxygen atoms bonded together.  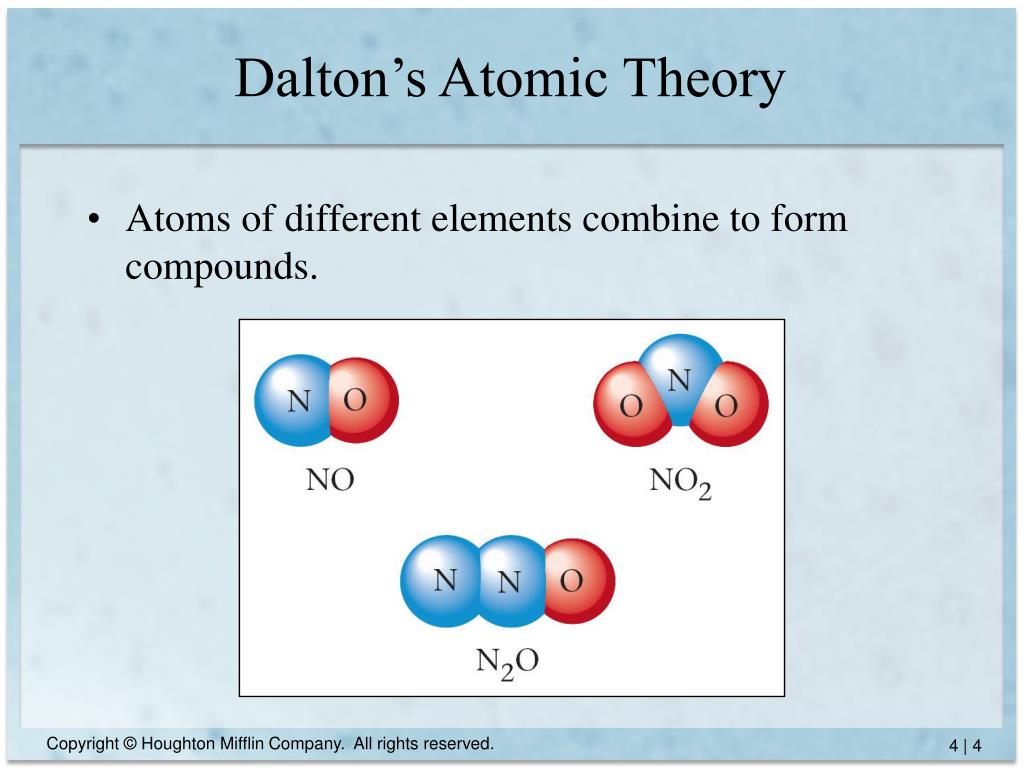 
The first postulate of Dalton’s atomic theory suggests that elements are composed of tiny indivisible particles called atoms. This concept was later put forth as the atomic theory by the English scientist John Dalton. Early Greeks like Democritus first advanced the idea of atomos, the smallest indivisible unit of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
